Expression of Ki-67 in Ovarian Tumors and its Correlation with Type, Grade and Stage
*Akhter S,1 Islam N,2 Kabir E,3 Begum S,4 Gaffar T,5 Khan AA6
| Abstract
Background: The identification of new proliferation markers could have clinical implications in ovarian carcinoma by stratifying patients for treatment and follow-up. Objectives: The study was conducted to evaluate the biological significance of proliferative marker Ki-67 antigen expression in benign and malignant ovarian tumors and correlate it with histological type, grade and stage of malignant tumor. Methods: The study was conducted in Sir Salimullah Medical College and Hospital. It was analytical cross-sectional study involving 100 cases of ovarian tumor (benign and malignant) over a period of two years. The proliferation expression related to Ki-67 antigen was evaluated by immunohistochemical monoclonal MIB-1 antibody. In each case, the Ki-67 labeling index (Ki-67 LI) was articulated as percentage of positively stained cells using high power objective of the microscope (x400). Results: Among the 100 cases of ovarian tumor, 50 were benign and 50 were malignant. Among the 50 benign tumor, 18 were serous, 9 mucinous, sex cord stromal tumor 8 and mature cystic teratoma 15 and in cases of malignant tumor 28 serous, 15 mucinous 1 on each of clear, endometrioid and transitional cell carcinoma. Total of 75% were grade II tumours, 22% grade I and 4% grade III. Ki-67 expression is high in malignant tumor than benign tumor. High Ki-67 LI was associated with high grade tumours (66.9%). Ki67 expression was also increase with increasing stage of cancer. CA 125 levels were significantly high in malignant tumor and did not have a significant correlation with Ki-67 LI. Conclusion: Ki-67 is an exceptionally a cost effective marker to determine the growth fraction of a tumour cell population. In ovarian tumor histological grade and stage when combined with Ki-67 LI in histopathology report would help in diagnostic differentiation of subtypes, prognostication, deciding the need for adjuvant chemotherapy and in predicting survival analysis.
|
[Journal of Histopathology and Cytopathology, 2019 Jan; 3 (1):15-26]
Keywords: Ovarian carcinoma, Proliferative index, Ki-67
- *Dr. Salma Akhter, Assistant Professor, Department of Pathology, Universal Medical College, Dhaka.
- Professor Dr. Nasimul Islam, Professor and Head, Department of Pathology, SSMC, Mitford Hospital. Dhaka.
- Professor Dr. Enamul Kabir, Professor, Department of Pathology, SSMC, Mitford Hospital. Dhaka.
- Shahanaz Begum, Associate Professor, Department of Pathology, SSMC, Mitford Hospital. Dhaka.
- Tania Gaffar, Assistant Professor, Department of Pathology, Uttara Adhunik Medical College. Dhaka.
- Abu Anis Khan, Consultant of Pathology, Sheikh Fazilatunnessa Mujib Memorial KPJ Specialized Hospital.
*For correspondence
Introduction
Ovarian tumors are a puzzling group of neoplasms that do not fall neatly into benign or malignant categories. Their behavior is enigmatic, their pathogenesis is unclear and their diagnosis, clinical management & prognosis is controversial.1 Ovarian cancer is the leading cause of death from gynaecological malignancies in the Western world and the highest incidence rates are found in North America and Northern and Western Europe.2 Over 70% of the women diagnosed with ovarian carcinoma have already advanced disease at the time of diagnosis.3 Ovarian cancer is the second most common gynaecological cancer worldwide and one of the leading cause of death due to malignancy in female3 and affects postmenopausal females in their sixties.4 The prognosis of ovarian cancer is poor, with an overall survival rate of about 40% in 5 years.3 Important prognostic factors include histological type, grade, stage of disease, age at diagnosis and the amount of residual disease after primary surgery.5,6 Furthermore, high proliferative activity in the ovarian tumor has been shown to imply poor prognosis.7,8 In tumor biology, proliferation has been recognized as a distinct hallmarks of cancer and act as an important determinant of cancer outcome. Cellular proliferation status of a tumor may be a diagnostic as well as prognostic tool.9 Mitotic count is the traditional and practical method of determining proliferative activity, but is hampered by several disturbing factors.10 Immunohistochemical detection of proliferating cells is an alternative way to determine the proliferative potential of a tumor and the expression of Ki-67 antigen has become a widely used marker.11 Ki-67 is a nuclear non histone protein that is present at low level in quiescent cells but increased in proliferating cells during G1, S, M, and G2 periods of cell cycle but absent in G0 phase.12 The expression of Ki-67 is associated with tumor aggression, prognosis, vascular invasion and tumor metastasis.11 It is an excellent marker to determine the growth fraction of a given cell population. The fraction of Ki-67 positive tumor cells is often correlated with the clinical course. MIB-1 is a commonly used monoclonal antibody on formalin fixed paraffin embedded tissue that detects the Ki-67 antigen.13,14 It is used in clinical application to detect the Ki-67 labeling index.11 High expression of Ki-67 has been found to indicate a poor prognosis in several cancer including ovarian cancer.15
The aim of this study was to compare the diagnostic significance of Ki-67 antigen expression in various type of ovarian tumor and correlation with histopathological grading and staging. So far known, no study has been done on expression of Ki-67 in ovarian tumor in Bangladesh.
Methods
This cross sectional study was carried out in the Department of Pathology, Sir Salimullah Medical College, Dhaka during the period of July 2014 to June 2016. Patients were enrolled from the Department of Gynaecology and Obstetrics, Sir Salimullah Medical College and Mitford hospital who having ovarian tumor and underwent surgical treatment. A total of 100 cases of ovarian masses were selected conveniently. Although the standard sample size was estimated to be 384 cases by using the formula n= z2pq(1-p)/d2, due to short study period and financial constrains, 100 cases were recruited. Histopathologically diagnosed primary ovarian tumors were included and nonneoplastic ovarian mass, like perovarian cysts, paratubal cysts, endometriotic cysts and also metastatic ovarian tumors were excluded in this study. The study has been permitted by the ethical review committee of Sir Salimullah Medical College. Permission for the study was also taken from the concerned departments from where study specimen were collected. All the necessary informations and clinical findings were recorded in a pre-structured proforma. All the cases were numbered chronologically and all the records, final histopathological reports and immunohistochemical reports were included in the proforma. After laparotomy, resected specimens were collected and preserved in 10% buffered neutral formalin solution at room temperature for 24 hours. After proper fixation, gross section was done and tissue were processed employing paraffin embedded method. One section was taken on a regular slide for haematoxyline and eosin stain and one section for immunostaining. Histopathological examination were done by light microscope at first in low power and then under high magnification. Histological typing and grading were done by WHO recommendation. Ki- 67 staining method. The corresponding blocks were cut into 3-5 micrometer thick sections on poly-l lysine coated slides. Ki-67 immunostaining were performed according to the manufacturer’s recommendations, using the MIB-1 clone as primary antibody and Envision as secondary antibody (Dako Carpenteria, CA & Ventana Medical systems, Tucson, AZ). Ki-67 immunostained slides were examined via light microscopy by two independent observers who were blinded to both the clinicopathological data and the results from the scoring for each section, positive Ki-67 staining were observed as brown, granular nuclear staining. For Ki-67 scoring the most positive area of the tumor was selected avoiding foci of inflammation. The number of positive nuclei was counted in 500 tumor cells in a high power field. The average of three counts over the same slide was taken and expressed as the percentage of Ki-67 positive cells in the tumor.
Results
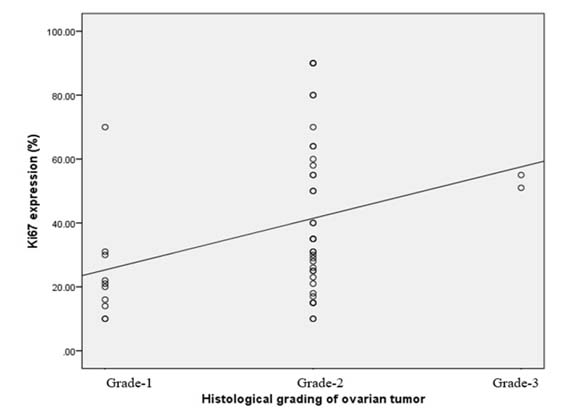
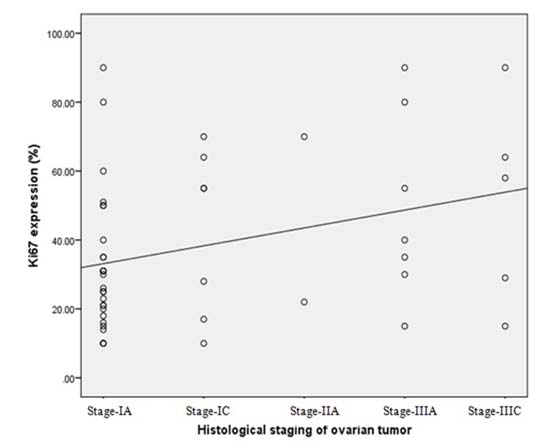
The mean age of the studied population was 38.39 years (SD ± 13.23)yrs (Table I). Ages of the study patients ranged from 15 to 70 years. The peak incidence of benign tumors was in 3rd decade of life while for malignant tumors was the 4th and 5th decades. Mean age of benign and malignant tumor was (33.50 ± 12.28) and (43.28 ± 12.42) respectively (Table II). The age of malignant tumor was significantly higher than benign. Family history of ovarian tumor was recorded (Table V). Out of 100 cases, twelve patients had positive family history of ovarian tumor. Of them, five cases were benign and seven cases were malignant. Forty one cases had no family history and forty seven cases could not give any history. Out of 50 cases of malignancy, ascitic fluid was available for cytological examination in 17 cases. Of them only five cases (29.4%) were found positive for malignant cells and 7 cases (70.6%) were found negative for malignant cells (Table IV). Total 100 cases of ovarian tumors, most of the cases had tumors of surface epithelial origin (73.0%). Next in frequency was germ cell tumors, followed by sex cord stromal tumors (8%). Out of 100 cases of benign and malignant ovarian tumors, CA 125 level was available in 49 cases, 8 cases were benign and 41 cases were malignant. Mean CA125 level in patients with benign and malignant tumor was(13.76 ± 6.65) and (222.68 ± 113.41) respectively (Table X). CA 125 level was significantly higher in malignant tumor than in benign (p<0.001). Ki-67 is a marker of proliferation, which expressed by the diving cells of a tissue. In this study, the percentage of Ki-67 expressing cells was seen in the ovarian tissue in both benign and malignant tumors. Mean Ki-67 expression in malignant tumor was (38.75 ± 23.61) and benign tumor was (1.69 ± 3.00) which was significantly higher in malignant compared to benign tumors (p< 0.001) (Table- VI). Ki 67 expression was correlated with tumor grading. It shows that increasing grade of tumor increase the Ki67 expression. Twenty two percent cases were of grade I and mean Ki-67 expression was 24.40 ± 16.68, 74.0% percent cases were of grade II mean Ki-67 expression was 43.64 ± 22.66 and finally 4.0% cases were of grade III mean Ki-67 expression was 53.00 ± 2.82 (Fig-1) . Correlation of histological grading with Ki-67 expression is shown in figure 1. An attempt was made to assign the malignant tumors into various pathological stages. 35 patients were in stage I, 9 patients were in stage II and the rests were in stage III at presentation. Most of the cases (54%) were of stage IA, 16% cases were of stage IIA, 14% cases were of stage IIIA and 12% cases were of stage IIIC (Fig-2). Ki-67 expression was increases with increasing stage of tumor (Fig-2).
Table I: Distribution of patients according to age (n=100)
| Age(years) | Frequency | % |
| ≤20 | 9 | 9.0 |
| 21 -30 | 26 | 26.0 |
| 31- 40 | 28 | 28.0 |
| 41- 50 | 20 | 20.0 |
| >50 | 17 | 17.0 |
| Total | 100 | 100.0 |
| Mean±SD | 38.39±13.23 | |
| Range (Min-Max) | 15 – 70 |
Table II: Distribution of patients according to benign and malignant tumor of ovary with age group (n=100)
| Type of tumor | n (%) | Age(years)
(Mean ± SD) |
p-value |
| Benign | 50 (50.0) | 33.50 ± 12.28 | <0.001s |
| Malignant | 50 (50.0) | 43.28 ± 12.42 |
Anova test was done to measure the level of significance.
s= significant
Table III: Distribution of patients according to family history of tumor (n=100)
| Family history of tumor | Benign | Malignant | (%) |
| Positive | 5 | 7 | 12 |
| Negative | 26 | 15 | 41 |
| Unknown | 19 | 18 | 47 |
| Total | 50 | 50 | 100 |
Table IV: Ascitic fluid for cytology in malignant tumors (n=17)
| Malignant cell | Frequency | % |
| Positive | 5 | 29.4 |
| Negative | 12 | 70.6 |
| Total | 17 | 100.0 |
Table V: Distribution of the study patients according to histopathological diagnosis(n=100)
| Histopathological type of tumor (n=100) | Benign
N=50 % |
Malignant
N=50 % |
||
| Surface epithelial tumor(73) | ||||
| Serous tumor (46) | 18 | 36 | 28 | 56 |
| Mucinous tumor (24) | 9 | 18 | 15 | 30 |
| Endometrioid tumor (1) | – | – | 1 | 2 |
| Clear cell tumor(1) | – | – | 1 | 2 |
| Transitional cell tumor(1) | – | – | 1 | 2 |
| Sex cord stromal tumor(8) | ||||
| Granulosa cell tumor | 4 | 8 | – | – |
| Fibroma | 4 | 8 | – | – |
| Germ cell tumor(19) | ||||
| Dysgerminoma | – | – | 2 | 4 |
| Mature cystic teratoma | 15 | 30 | – | – |
| Mixed germ cell tumor | – | – | 1 | 2 |
| Yolk sac tumor | – | – | 1 | 2 |
| Total 100 | 50 | 100 | 50 | 100 |
Table VI: Distribution of CA-125 in benign and malignant ovarian tumor (n=49)
| Tumor type | CA-125 | |
| Mean±SD Range | ||
| Benign | 13.75 ±6.65 | 6.19 – 25.0 |
| Malignant | 222.68 ± 113.41 | 11.50 – 501.0 |
| P value | <0.001s
|
|
Mann Whitney U test was done to measure the level of significance
Table VII: Relation of Ki-67 expression in benign and malignant ovarian tumor (n=100)
| Tumor type | Ki-67 expression(%) | P value | |
| Mean ±SD | Range | ||
| Benign | 1.69 ±3.0 | 0-5 | <0.001 |
| Malignant | 38.75 ±23.61 | 10-90 | |
Figure 1. Correlation of histological grading of ovarian tumor with Ki-67 expression. Here a positive significant relation is achieved (rs=0.374, p<0.009).
Figure 2. Correlation of histological staging of ovarian tumor with Ki 67 expression.
This figure shows Positive correlation between histological staging of ovarian tumor and Ki-67 expression.
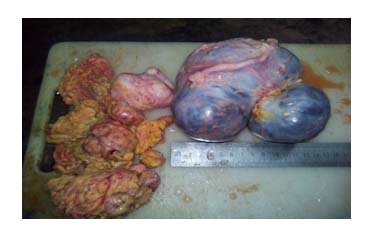
Figure 3. Photograph of mucinous cyst adenocarcinoma
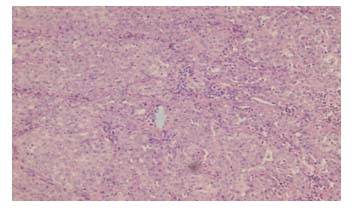
Figure 4. Photomicrograph of mucinous cyst adenocarcinoma in H&E staining (x40)
Figure 5. Photomicrograph of serous cystadenocarcinoma in H&E staining (x40)
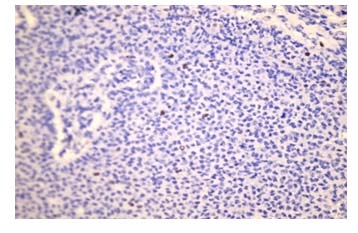
Figure 6. Photomicrograph of serous cystadenocarcinoma in immuno staining by Ki-67 (x40)
Figure 7. Photomicrograph of granulose cell tumor H&E staining(x20)
Figure 8. Photomicrograph of granulose cell tumor in immunostaining by Ki-67(x20)
Figure 9. Photomicrograph of dysgerminoma in H&E staining (x20).
Figure 10. Photomicrograph of dysgerminoma in immunostaining by Ki-67(x40).
Discussion
Ovarian cancer is the third most common malignancy among women accounting for 5.5% of all cancers.16 About 80% of all ovarian tumors are benign and occur mostly in young women between 25 and 40 years of age. The importance to distinguish benign and malignant tumor in reproductive age group where the conservation of other ovary is important. Over 70% of the women diagnosed with ovarian carcinoma have advance disease at the time of diagnosis. Important prognostic factors include stage of disease, age at diagnosis, histological type and grade17. Furthermore, high proliferative activity in the ovarian tumor has been shown to imply a poor prognosis18 . Mitotic count is a traditional and practical method to determine proliferative activity, but hampered by several disturbing factors. Ki-67 is an easily available, more economical, rapid, used on formalin fixed paraffin embedded sections and a more reproducible biomarker available in developing countries, compared with other markers like proliferating cell nuclear antigen and bromodeoxy uridine. Ki-67 immunostaining requires only small tissue samples, allowing it to be applied even in case of patients who are candidates for Neo Adjuvant Chemotherapy (NACT)
Histopathological diagnosis is routinely done on by formalin fixed paraffin embedded tissue followed by microscopic analysis of H&E stained section. However, this approaches may occasionally fail to precisely diagnose or provide adequate prognostic information.19 It is therefore important to find new and reliable cellular markers that can overcome these limitations.
Uncontrolled cellular proliferation is one of the most important biological mechanisms involved in onchogenesis.20 The introduction of biomarkers could facilitate a simpler and more consistent method to determine the proliferation potential of a tumor. This could have clinical implication, both in indicating prognosis and tailoring chemotherapy. Immunohistochemical detection of proliferating cells is a way to determine the proliferative potential of a tumor.21 Ki-67 is the most studied proliferation marker in cancer research which is overexpressed in malignant tissues compared to benign tissues. Furthermore, high proliferative activity in the ovarian tumor has been shown to imply a poor prognosis.22,23
Present study was undertaken to find out the value of Ki67 expression in the diagnosis of benign and malignant tumor and the relation of ki67 expression with the grading and staging of malignant tumor.
A total 100 cases were included in this study. 50 cases of benign and 50 cases of malignant ovarian tumor were taken and nonovarian lower abdominal masses (perovarian cyst, paratubal cyst etc) were excluded from the study.
In present study, mean age of the patients with ovarian tumor was 38.39 ± 13.23 years with the range of 15- 70years. Most of the patients were more than 30 years old (Table I) and highest number of malignant cases were seen in the 4th to 5th decade (Table II). Dhar et al. (2015) found mean age 38.2 ±7.31 years with a range of 16 to 69 years and maximum patients were more than 30 years old, which was almost similar to present study.24
Age was significantly higher in patient with malignant tumor compared to patient with benign tumor according to Baloch S et al. 2008. In present study, mean age was 33.50 ± 12.28 in patients with benign tumor and 43.28 ± 12.42 in malignant tumor patient (Table II) which correlate with the study of Baloch et al. The risk of developing ovarian cancer gets higher with age(American Cancer Society, 2015).25
Among study population (n=100) 38.0% had regular and same number had irregular menstruation, 24.0% of them were menopausal (Table IV).
Regarding family history of ovarian tumor 12.0% of patients were found to have positive family history of ovarian tumor in the present study (Table V).
Out of the 50 patients with malignant ovarian tumor ascitic fluid for cytology were available in 17(34.0%) cases. And only five cases (29%) were found to contain malignant cell. A study of 276 ascitic cytology specimens 48 (17.0%) were found to contain malignant cells.26
Regarding histopathological diagnosis of tumor type, surface epithelial tumor were 73.0%, sex cord stromal tumor were 8.0% and germ cell tumor were 19.0%. Among surface epithelial tumor serous type of tumor was maximum 46 (28.0% were serous cyst adenocarcinoma followed by 18.0% serous cyst adenoma), than mucinous tumor 24 (15.0% mucinous cyst adenocarcinoma, and 9.0% mucinous cyst adenoma), endometrioid carcinoma 1, transitional carcinoma 1 and clear cell carcinoma 1. Among 8 of sex cord stromal tumor, 4 were of granulosa cell tumor and 4 were of fibroma and in case of germ cell tumor, benign mature cystic teratoma 15, malignant dysgerminoma 2 and 1 mixed germcell tumor and 1 yolk sac tumor.
According to Robins and Cotran serous neoplasm were account for approximately 40%, mucinous for about 20-25%, endometrioid 10% of all ovarian neoplasm. Rajagopal IK and Ramesh (2016) found serous cyst adenocarcinoma 29.1 % and mucinous cystadenocarcinoma 32.4%. In this study 28% of serous cystadenocarcinoma and 15% of mucinous cystadenocarcinoma were found.
Regarding tumor marker expression CA-125, it was obtained in 49 cases and the mean obtained in benign tumor compared to malignant tumor was statistically significant (p = <0.001). Mean CA-125 was (222.68 ± 113.41) in malignant and (13.76 ± 6.65) in benign in the present study. Baloch et al. (2008) found CA125 normal in 79.3% and raised in 20.68% patients with ovarian tumor.27
In the present study Ki-67 expression in benign tumors were found to have lower number of positive case (1.69 ± 3.00) compared to malignant tumor (38.75 ± 23.61). On statistical analysis the difference of Ki-67 expression between benign and malignant tumor were found to be significant (p<0.001). Choudhury et al. (2011) revealed that mean Ki-67 index was 3.2 ± 3.7 in benign tumors while malignant tumors had a mean Ki-67 index of 33.1 ± 16.7, the difference was statistically significant which was almost similar to this result.28 Rajgopal and Ramesh found mean Ki 67 2.9% in benign and 29.9% in malignant tumors.
Regarding grading of malignant ovarian tumor, 22.0% cases were of grade I (mean Ki-67 expression was 24.40 ± 16.68), 74.0% percent cases were of grade II (mean Ki-67 expression was 43.64 ± 22.66) and finally 4.0% cases were of grade III (mean Ki-67 expression was 53.00 ± 2.82). There were significant differences among the groups. When compared between grade I & grade II and between grade I and grade III they were also found to be statistically significant.
In this study, histological grading had a significant correlation with Ki-67 expression (Figure 1) of patients with ovarian tumor. That means increase of grading causes Ki-67 expression increase. However, Ki-67 expression was also positively correlated with tumor grade in a study by Rajagopal and Ramesh, (2016).
When the malignant ovarian tumors of ovary were staged, maximum (54.0%) cases were of stage IA (mean Ki-67 expression was 33.25 ± 19.37), 16.0% percent cases were of stage IC (mean Ki-67 expression was 44.25 ± 22.61), 4.0% cases were of stage IIA (mean Ki-67 expression was 46.00 ± 33.94), 14.0% cases were of stage IIIA (mean Ki-67 expression was 49.28 ± 27.29) and 12.0% cases were of stage IIIC (mean Ki-67 expression was 51.16 ± 25.10). Though malignant tumor of ovary with high stage caused higher expression of Ki-67 there was no significant difference of Ki-67 expression among the different stages.
In this study, significant positive correlation was found between histological staging of ovarian tumor and Ki-67 expression (Fig 2). Choudhury et al. (2011) found significant correlation between the Ki-67 index and stage of the tumor.28 Similarly, Rajgopal and Ramesh, (2016) revealed positive correlation between Ki-67 expression and tumor stage.
Limitations
The study was done in limited time of span, respondents were collected from only one center hence it may not represent the whole population of the country. Borderline tumors were not included in this study. The sample size was small. The limitation of the study is inability to follow up the patients and assess the prognosis.
Conclusion
Ki-67 expression showed significant difference between benign and malignant tumor and positive correlation with histopathological grading and staging. Ki-67 and CA-125 expressions were more obvious in malignant tumors compared to benign tumors. Ki-67 expression was increase with increasing grading as well as staging. SEOC have the worst prognosis among the gynecological malignant tumours. A number of factors are known to persuade survival in ovarian cancer, till date, histologic grade and FIGO stage for diagnosis are considered the most important for prognostication. Ki-67 LI help in diagnostic and prognostication by differentiation of the morphologic types when collectively studied with architectural grade and FIGO stage, predicting the response to chemotherapy and overall survival of the patient. In recent times, it is also a potentially remarkable therapeutic target. MIB 1 antibody against Ki-67 antigen is widely available and a cost effective investigation, especially in a developing country where SEOC are on the rise. Ki-67 LI IHC marker should be included in histopathology report regularly as a diagnostic and prognostic factor there by it will pay the way for better understanding of biological behaviour and modify treatment strategies.
Recommendation
Study period should be extended. Further multi centered prospective study with large sample size should be recommended.
References
- Rajagopal L and Ramesh S. Immunohistochemical expression of Ki-67 in ovarian tumors & correlation with clinocopathological factors. Int J Pharm Bio Sci. 2016; 7(1), 67-73.
- Tavassoli FA, Devilee P. In pathology and Genetics of tumours of Breast and Female Genital Kleihuses P, Sobin LH, editors. Lyon: International Agency for Research on Cancer; 2003.
- Jemal A, Bray F, Center, MM, Ferlay J. Ward E, and Forman D. Global cancer statistics. CA Cancer Journal for Clinicians, 2011; 61(2): 69-90.
- K Uma Devi. Current status of gynecological cancer care in India,” Journal of Gynecologic Oncology, 2009; 20 (2): 77–80.
- Armstrong DK and Brady M. Intraperitoneal therapy for ovarian cancer: a treatment ready for prime time. J Clin Oncol, 2006; 24: 4531-4533.
- Clark TG, Stewart ME, Altman DG, Gabra H and Smyth JF. A prognostic model for ovarian cancer. Br J cancer, 2001;85:942-952
- Garzetti GG, Ciavattini A, Goteri G, Denictolis MG et al. Ki67 antigen immunostaining (MIB-1 monoclonal antibody) in serous ovarian tumors,index of proliferative activity with prognostic significance. Gynecol Oncol,1995; 56:169-174.
- Sengupta S, Mc Gown AT, Bjaj V, Blackhall F, Swinder R, Bromly M et al, P53 and related proteins in epithelial ovarian cancer. Eur J Cancer, 2003;36: 2317-2328.
- Linden MD, Kubus J and Zarbo RJ. Clinical application of morphological and immunocytochemical assessments of cell proliferation. Am J Clinpathol, 1992;9: 4-13.
- Guro Aune, Astrid K. Stuns Salveig Tingulstad, Qyvindsalvesen et al. The proliferation marker Ki-67/MIB- 1 PPH3 and Survivin.2011;
- Gerdes J, Schwab U, Lemke H, et al. Production of a mouse monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer,1983; 31:13-19.
- Korkolopoulou P, Vassilopoulos I, Konstantinidou AE, Zorzos H et al. The combined evaluation of p27 Kip1 and Ki-67 expression provides independent information on overall survival of ovarian carcinoma patients. Gynecol Oncol, 2002; 85(3):404–414.
- Gotlieb WH, Goldberg I, Weisz B et al. Topoisomerase II immunostaining as a prognostic marker for survival in ovarian cancer.Gynecologic Oncology, 2001;82 (1), 99–104.
- Khouja MH, Baekelandt M, Nesland JM and Holm R. The clinical importance of Ki-67, p16, p14, and p57 expression in patients with advanced ovarian carcinoma. Int J GynecolPathol, 2007;26(4):418–425.
- Pollack A, DeSilvio M, Khor LY, Al-Saleem TI et al. Ki-67 staining is a strong predictor of distant metastasis and mortality for men with prostate cancer treated with radiotherapy plus androgen deprivation: Radiation Therapy Oncology Group Trial 92-02. J ClinOncol, 2004; 22: 2133–2140.
- Gupta MC and Mahajan BK. In Epidemiology of non-communicable diseases, Textbook of Preventive and Social Medicine, 2003; 3rd Edn.: Jaypee Brothers, 306-328.
- Kumar V, Abaasm AK and Faustom N. Robbins and Cotran Pathologic Basis of Disease, 2004; 7th Edn, WB Saunders Company, Philadelphia,1059-11.
- Armstrong DK and Brady M. Intraperitoneal therapy for ovarian cancer: a treatment ready for prime time. J Clin Oncol, 2006; 24: 4531-4533.
- Egan MJ, Crocker J. Significance of silver stained nucleolar organizer regions in benign breast disease and breast cancer. Natl Cancer. Inst.1992.
- VanDiest PJ, Brugal G and Baak JP. Proliferation markers in tumors: interpretation and clinical value. J Clinpathol 1998;51(10):716-724.
- Christopher DM. Diagnostic Histopathology of Tumors. Churchill Livingstone, 2007; 567-576.
- Garzetti GG, Ciavattini A, Goteri G, Denictolis MG. Ki-67 antigen immunostaining ( MIB1 monoclonal antibody) in serous ovarian tumor : index of proliferative activity with prognostic significance. Gynecol Oncol. 1995; 56: 169-174.
- Sengupta PS, Mc.Gown AT, Bjaj V, Blackhall F, Swinder R, Bromly M,et al. p53 and related proteins in epithelial ovarian cancer. Eur J cancer. 2000; 36:2317-2328.
- Dhar SR, Begum SN, Zabin F and Akhter S. Socio-demographic Characteristics of Ovarian Tumor Patients attended at a tertiary Care Hospital in Dhaka city. J Curr Adv Med Res, 2015; 2(2):39-41.
- American Cancer Society. What are the risk factors for ovarian cancer? Available at: http://www.cancer.org/cancer/ovariancancer/detailedguide/ovarian-cancer-risk-factors. 2015;
- Karoo RO, Lioyd TD, Garacea G, Redway HD. How valuable is ascitic cytology in the detection and management of malignancy. Postgrad Med J, 2003;79:292-294 .
- Baloch S, Khaskheli M, Malik AM, Sheeba A and Khushk IA. Clinical spectrum and management of ovarian tumors in young girls upto 20 years of age. J Ayub Med Coll Abbottabad, 2008;20(4):14-17.
- Choudhury M, Goyal S and Pujani M. A cytohistological study of Ki-67 expression in ovarian tumors. Indian J Pathol Microbiol, 2011; 54(1): 21-22
- Rosai J .Rosai and Ackerman’s surgical pathology 10th edition, Female reproductive system, Elsevier, St. Luis Missouri, 2011;19 (2):1649-1736.