The Role of Special Stains in Trephine Biopsy of Lymphoma
*Yesmin I,1 Begum F,2 Yunus A,3 Kabir AL,4 Baqui MN5
*Dr. Israt Yesmin, Specialist Pathology, Pathology and Laboratory Medicine, Square Hospital, Dhaka. badhanydr29@gmail.com
- Ferdousy Begum, Associate Professor, Department of Pathology, Bangabandhu Sheikh Mujib Medical University, Dhaka. ferdousy_begum2000@yahoo.com
- ABM Yunus, Professor & Chairman, Department of Haematology, Bangabandhu Sheikh Mujib Medical University, Dhaka. abmyunus.bsmmu@gmail.com
- Amin Lutful Kabir, Associate Professor, DEPT of Haematology, BSMMU Email aminlutful@gmail.com
- Muhammad Nazmul Baqui, Senior Lecturer, Faculty of Medicine, AIMST University, Kedah, Malaysia. doctornazmul@yahoo.com
*For correspondence
Abstract
Background: Accurate diagnosis of lymphoma is always challenging to histopathologists. Aside from routine H& E stain special stains play an important role inreaching the final diagnosing of lymphomacases. Here we have studied the use of these special stains in trephine biopsy material of lymphoma cases.
Methods: This descriptive cross sectional study was carried out at the Department of Pathology Bangabandhu Sheikh Mujib Medical University (BSMMU), Dhaka, from July 2012 to June 2014. Total 17 cases, which underwent trephine biopsy were included in this study. Different special stains and immunostains were done to reach the final diagnosis. Statistical analyses were performed to find out the usefulness of these special stains.
Results: Fourteenpatients were male and rests were female. Mean age of the patients was 41.8±12.5 years. IHC of trephine biopsy material aids in diagnosis of lymphomatous infiltration with further sub classification. While conventional H&E and giemsa stain revealed only 6(35.3%) cases with lymphomatous infiltration of bone marrow. Among these 14 cases, 10 (58.8%) cases were B-NHL, 3 (17.6%) cases were T-NHL and rest 1(5.9%) was lymphocyte-rich classical Hodgkin lymphoma.
Conclusion: It was found that, use of special stains and IHC of trephine biopsy material of lymphoma cases improved the diagnostic accuracy of lymphoma.
[Journal of Histopathology and Cytopathology, 2019 Jul; 3 (2):134-142]
Key words: Trephine biopsy
Introduction
Trephine biopsy of the bone marrow has wide application in clinical medicine. Its important use is in the evaluation of patients with various haematological disorders like malignant lymphoma, acute leukaemias, myeloproliferative disorders, myelodysplastic syndromes, metastatic tumor, granulomatous disorder, myelofibrosis, aplastic anaemia and plasma cell dyscrasias. Examination of the bone marrow aspirate and trephine biopsy is essential for the diagnosis of bone marrow disorders. The aspirate and trephine biopsy provide complimentary and useful information. It is recommended that both bone marrow aspirate and biopsy be routinely performed for correlation of representative findings.
The bone marrow trephines are particularly useful in identifying focal lesion that may be less apparent on aspirate smear alone. The bone marrow biopsy is also a common and important staging procedure that is performed routinely on patients with hematologic and non-hematologic malignancy. Pathomorphological examination of trephine biopsies of the bone marrow represents a standard method for the diagnosis and staging of hematological neoplasms and other disorders involving the bone marrow. The use of special stains in bone marrow pathology should be determined following examination of the routinely stained biopsy and the patient’s clinical history.1 Although conventional morphology remains the gold standard for paraffin embedded BM trephines, IHC staining has become an integral part of the diagnostic and prognostic workup.2 Distinction of the most common type of B-NHL in the trephine biopsy can be difficult on morphological ground alone due to overlapping cytological features and distribution pattern and therefore require IHC and occasionally molecular data for primary diagnosis.2
Non Hodgkin lymphoma (NHL) composed of small B lymphocytes comprises the majority of lymphoma infiltrates in the bone marrow. Bone marrow involvement is seen in virtually 100% of B cell chronic lymphocytic leukemia (B-CLL) / small lymphocytic lymphoma (SLL), in almost all cases of lymphoplasmacytic lymphoma (LPL) and hairy cell leukemia (HCL), in 55-93% of mantle cell lymphoma (MCL), in 50-60% of follicular lymphoma (FL) and invariably in splenic marginal zone B cell lymphoma (SMZL). In nodal and extra nodal marginal zone B cell lymphoma the marrow involvement is found in 10-30% cases.2
The pattern of bone marrow involvement is one of the most important hints for diagnosis. Different patterns of bone marrow infiltration by NHLs are recognized, namely paratrabecular, randomly focal / intertrabecular / non paratrabecular, interstitial, diffuse and sinusoidal / intravascular. In some cases, multiple or mixed pattern are present in the same specimen. Although most infiltrates are easily recognizable in standard stains, sparse interstitial and sinusoidal involvement can be practically undetectable by conventional morphology and requires IHC to highlight the neoplastic population.2
Lymphoma demonstrate significant increased incidence rates throughout the world and causes mortality.3 Though uses of special histochemical stain in trephine biopsy is an established practice in histopathology, the application of immunohistochemistry (IHC) to diagnostic bone marrow trephine biopsy specimen is a relatively new practice.4 IHC is a powerful auxiliary technique for the diagnosis of hematologic disorder in bone marrow trephine biopsies.2 IHC reveals clear architectural features and estimates tumor cellularity in paraffin sections. By using IHC an accurate characterization and primary classification of lymphoid neoplasm is possible in the majority of cases and expands the diagnostic scope of bone marrow histology.2 The ability to define such subgroups is increasingly important as treatment is tailored not only to specific types of lymphoma but also on the basis of their prognostic group.5 Therefore the aim of this study was undertaken to see the role of special stains in bone marrow trephine biopsy in the diagnosis of lymphoma and to detect lymphoma of bone marrow and their typing.
Methods
This descriptive cross sectional study was carried out at the Department of Pathology Bangabandhu Sheikh Mujib Medical University (BSMMU), Dhaka, from July 2012 to June 2014. A total of 17 patients of different age and sex were selected from the department of Hematology BSMMU, Dhaka and other different institutes of Dhaka by using non probability sampling method. Patients who underwent trephine biopsy and histologically and clinically suspected as cases of lymphoma and cases accompanied by clinical information were included in this study. Exclusion criteria include patients who were not suspected as cases of lymphoma in histological diagnosis, specimen having severe crush artifact and specimen less than 1 cm in length. According to standard protocol the biopsy samples were taken from the posterior superior iliac spines under local anesthesia. Formalin fixed trephine biopsy sample were allowed to fix over night at room temperature in 10% buffered formalin fixative. Trephine biopsy samples collected from haematology department of BSMMU and other different institutes of Dhaka were decalcified in 10% formic acid for four hours followed by wash in running tap water for an hour. Routine processing and paraffin embedding were done according to BSMMU standard tissue processing protocol in automated machines. Paraffin blocks were made. Subsequent tissue sections were analyzed by using routine H&E stain, periodic acid Schiff (PAS) stain, Giemsa stain and a primary histological diagnosis were made.
Sections with at least five well preserved marrow spaces were studied for cellularity, normal hematological elements, presence of infiltration if any, the extent, histologic pattern and morphology of infiltration and other secondary changes. The morphology and histologic pattern of infiltration were categorized as diffuse, interstitial, focal (non-paratrabecular), paratrabecular and mixed patterns. Diffuse pattern was defined when there was extensive replacement of the marrow elements, both hematopoietic tissue and fat, obscuring the marrow architecture. Interstitial pattern was indicated when individual neoplastic cells were interspersed between hematopoietic cells and fat, focal, where nodular aggregates were seen separated by normal hematopoietic marrow, paratrabecular, when infiltrate aggregates were seen immediately adjacent to bony trabeculae.
2–3 μm thick sections were cut from the paraffin blocks for IHC. EnVision method was followed. The immunohistochemical staining procedure of all antibodies except TdT was performed in the IHC lab of BSMMU by following its staining protocol. Table I shows the IHC panel done for diagnosis.
Results of immunostains were assessed by light microscopy using a 10× objective lens with subsequent use of 20 and 40 lenses for confirmation. Immunostains results were expressed as positive, weakly positive or negative. Cases where quantification of specific type of immunohistochemical markers positive cells are needed counted at x40 magnification, excluding cortical and trabecular bone, periosteal connective tissue, adipose tissue or areas of haemorrhage. The specific immunohistochemical marker positive cells were counted in five or more randomly selected fields and the number of positive cells was expressed as a percentage of total number of bone marrow nucleated cells. Slides were reviewed by two senior histopathologists. Subsequently special histochemical stains and immunohistochemical finding was analyzed and final reports were issued. This was performed in the department of pathology of BSMMU, Dhaka.
Results
Age, sex and clinical findings
It was observed that maximum case (35.3%) patients belonged to age grouped 31-40 years. The mean age was found 41.8±12.5 years with range from 20 to 70 years. Majority14 (82.4%) patients were male and 3(17.6%) were female indicating male predominance. Male and female ratio was 4.7:1. Out of 17 patients of the present study 13 patients had fever, 6 complained of weakness and 6 presented with hemorrhagic manifestation e.g. gum bleeding, cyanosis, bleeding spot etc. The other complains included weight loss, bone and joint pain, swelling of whole body, night sweat, pruritus, yellow discoloration of skin and sclera, neck swelling, oral ulcer etc. Anaemia was the most prevalent (88.2%) feature. Other signs included jaundice, oedema, cyanosis etc. Five (29.4%) patients had generalized lymphadenopathy, two (11.8%) had grouped palpable lymph node, one (5.9%) had bony tenderness and all had normal tonsil.
Ultrasonographic findings
Among 17 cases USG reposts were not available in 5 cases. Two (11.8%) patients had splenomegaly, 2(11.8%) had hepatomegaly, 6(35.3%) had hepatosplenomegaly, 1(5.9%) had abdominal lymphadenopathy and USG findings were normal in 1(5.9%) cases.
Comparison of Primary histological diagnosis with final diagnosis after IHC
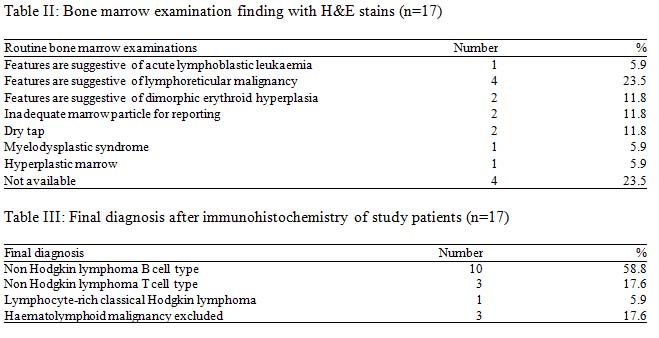
Out of these 17 cases 14 cases as various types of lymphoma. In the present study, primary histological diagnosis on trephine biopsy was made as haematolymphoid malignancy unclassified in 11(57.1%) cases and NHL in 6(42.9%) cases. Fibrosis was not seen in any of the 17 cases.

Trephine biopsy samples of 17 cases were histologically analyzed with the help of H&E stain along with PAS and Giemsa stain. Five patients were finally diagnosed as NHL by primary histological diagnosis was made on the basis of different pattern of abnormal cellular infiltration in the bone marrow trephine biopsy.
Discussion
In this series it was observed that most of the patients with haematological disorder were in 4th decade and their mean age was 36.1±15.01 years. Male to female ratio was almost 3:1. Similarly study conducted by Chen et al. (2000) observed male to female ratio was 2.9:1.6 Similar observations regarding the male predominant were also made by Matsuo et al. (2003) and Lone & Naeem (2011).7 On the other hand Kumar et al. (2009) observed female predominant, where they found female to male ratio was almost 5:1.8
The pattern of marrow involvement also differs to some extent in various NHL subtypes, for example paratrabecular infiltration is strongly associated with follicular lymphoma. A bone marrow biopsy performed in patients with low grade lymphoma sometime shows unexpected high grade transformation, which necessitates a different therapeutic approach.9
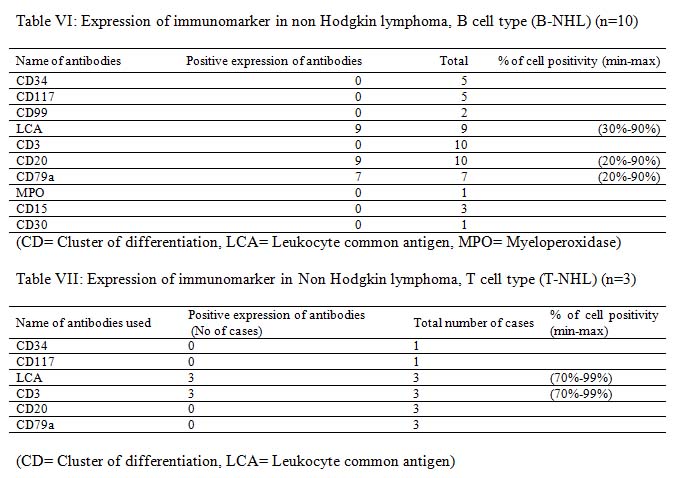
Among initial 11 cases of haematolymphoid malignancy unclassified in trephine biopsy were finally seven of them were diagnosed as B-NHL and one case as Hodgkin lymphoma. The remaining three cases were finally excluded from haematolymphoid malignancy. This result indicates the importance of IHC in the diagnosis of haematolymphoid malignancy. The remaining six cases of NHL were further sub classified as B-NHL and T-NHL (Table IV).
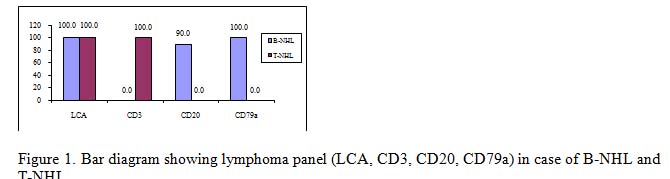
It is very much clear that trephine biopsy reports are superior as it could specify the diagnosis in most of the cases with the exception of cases of B-NHL and T-NHL. The latter cases were diagnosed only on the basis of CD3, CD20 and CD79a immunomarker. The incidence of bone marrow involvement in extra nodal MZL is lower (~20%) than in other low grade B-NHL, albeit recent studies described BM infiltration in up to 44% of cases.10
West et al. (2002) studied 61 cases (26 follicular lymphoma and 35 benign or atypical aggregates). They found that no single stain is sufficient for identification of neoplastic lymphoid aggregates. However, this distinction was made possible by using a panel of antibodies. The most useful antibodies were CD10, bcl-2, CD5, and CD20. Most benign or atypical aggregates do not express CD10 and CD23.11
Bone marrow specimens from 317 patients with non-Hodgkin’s lymphoma (NHL) obtained at initial staging was evaluated for the presence of lymphoma or benign lymphoid aggregates were studied by Conlan et al. (1990). They have found thirty-two percent (102 patients) had lymphoma in their bone marrow, and 9% had benign lymphoid aggregates. Bone marrow lymphoma was present in 39% of low-grade, 36% of intermediate-grade, and 18% of high-grade lymphomas. The bone marrow was involved in 25% of patients with diffuse large-cell or immunoblastic NHL. Bone marrow involvement did not affect survival of patients with low-grade NHL, but survival was significantly shorter (P<0.05) for patients with intermediate- and high-grade NHL with bone marrow involvement. Bone marrow involvement was equally common in B-cell and T-cell NHL (31% v 32%).12
Bone marrow involvement by lymphoma is considered a systemic dissemination of the disease arising elsewhere, although some tumors may arise primarily in the bone marrow microenvironment 13. IHC is important for lymphoma in particular those with heterogeneous histology to establish accurate diagnosis and adequate therapy14.
Bone marrow aspiration and bone marrow biopsy are important diagnostic procedures for diagnosis of hematological, non-hematological malignancies and other diseases. These procedures are also valuable for follow up of patients undergoing chemotherapy, bone marrow transplantation and other forms of medical treatment. In the study conducted by Toi et al. (2010), comparative evaluation of all such bone marrow aspiration (BMA) and bone marrow biopst (BMB), to see the complementary role of both the procedures, to study the advantages and disadvantages of both the procedures done simultaneously. There was 61.25% positive correlation between BMA and BMB in the study15.
Conclusion
It can be concluded that correct diagnosis can be made in majority of cases with the application of PAS, Giemsa and limited number of immunomarker. Correct and relevant clinical information along with appropriate special stain of bone marrow trephine biopsy improves the diagnostic accuracy of lymphoma cases.
References
- Brunning RD, Arber DA, 2011. Bone marrow. In J, Rosai Rosai and Ackerman’s Surgical Pathology, Tenth edition, Mosby Elsevier Inc, pp. 1927-2012.
- Kremer M, Quintanilla-Martínez L, Nährig J, von Schilling C, Fend F. Immunohistochemistry in bone marrow pathology: a useful adjunct for morphologic diagnosis. Virchows Arch Int J Pathol. 2005 Dec;447(6):920–37.
- Tahmasby B, Marnani AB, Maleki M, Barouni M, Mousavi SH, Naseriyan B, et al. Blood malignancies in Mazandaran province of Iran. Asian Pac J Cancer Prev APJCP. 2013;14(2):1053–6.
- Olsen RJ, Chang C-C, Herrick JL, Zu Y, Ehsan A. Acute leukemia immunohistochemistry: a systematic diagnostic approach. Arch Pathol Lab Med. 2008 Mar;132(3):462–75.
- Guidelines on the management of acute myeloid leukaemia in adults – – 2006 – British Journal of Haematology – Wiley Online Library [Internet]. [cited 2019 Sep 30]. Available from: https://onlinelibrary.wiley.com/doi/full/10.1111/j.1365-2141.2006.06314.x
- Clinicopathological studies on bone marrow involvement of non-Hodgkin’s lymphoma [Internet]. ResearchGate. [cited 2019 Sep 30]. Available from: https://www.researchgate.net/publication/12095478_Clinicopathological_studies_on_bone_marrow_involvement_of_non-Hodgkin’s_lymphoma
- Matsuo T, Kuriyama K, Miyazaki Y, Yoshida S, Tomonaga M, Emi N, et al. The percentage of myeloperoxidase-positive blast cells is a strong independent prognostic factor in acute myeloid leukemia, even in the patients with normal karyotype. Leukemia. 2003 Aug;17(8):1538–43.
- Kumar S, Rau AR, Naik R, Kini H, Mathai AM, Pai MR, et al. Bone marrow biopsy in non-Hodgkin lymphoma: a morphological study. Indian J Pathol Microbiol. 2009 Sep;52(3):332–8.
- Juneja SK, Wolf MM, Cooper IA. Value of bilateral bone marrow biopsy specimens in non-Hodgkin’s lymphoma. J Clin Pathol. 1990 Aug;43(8):630–2.
- Feugier P, De March AK, Lesesve JF, Monhoven N, Dorvaux V, Braun F, et al. Intravascular bone marrow accumulation in persistent polyclonal lymphocytosis: a misleading feature for B-cell neoplasm. Mod Pathol Off J U S Can Acad Pathol Inc. 2004 Sep;17(9):1087–96.
- West RB, Warnke RA, Natkunam Y. The usefulness of immunohistochemistry in the diagnosis of follicular lymphoma in bone marrow biopsy specimens. Am J Clin Pathol. 2002 Apr;117(4):636–43.
- Conlan MG, Bast M, Armitage JO, Weisenburger DD. Bone marrow involvement by non-Hodgkin’s lymphoma: the clinical significance of morphologic discordance between the lymph node and bone marrow. Nebraska Lymphoma Study Group. J Clin Oncol Off J Am Soc Clin Oncol. 1990 Jul;8(7):1163–72.
- Primary bone marrow lymphoma: an uncommon extranodal presentation of aggressive non-hodgkin lymphomas. – PubMed – NCBI [Internet]. [cited 2019 Sep 30]. Available from: https://www.ncbi.nlm.nih.gov/pubmed/22251943
- Bone marrow aspiration before bone marrow core biopsy using the same bone marrow biopsy needle: a good or bad practice? [Internet]. [cited 2019 Sep 30]. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1860622/
- Toi PC, Varghese RG, Rai R. Comparative evaluation of simultaneous bone marrow aspiration and bone marrow biopsy: an institutional experience. Indian J Hematol Blood Transfus Off J Indian Soc Hematol Blood Transfus. 2010 Jun;26(2):41–4.